Many parents believe that vaccine solution is sterile and contains a weakened virus, salt water and a little preservative. This is what I used to think prior to becoming pregnant with my first child.
In 1995, when I was researching vaccinations in order to make a decision for my expected baby, I telephoned the Vegan Society to ask if they could confirm that the tetanus toxoid componant of the DPT was manufactured using horses blood.
The lady on the other end of the phone exclaimed
"Oh, I don't think so because that would be unsanitary!"
What unsuspecting consumers don't realise is the 'science' of
vaccination is crude and anything but sanitary.
Animal products such as monkey kidney cells, horses blopd, chick embryo, aborted human fetal lung and eye tissue and human blood products are involved in the culture of viruses and bacteria and are present in vaccines in trace amounts and listed in the vaccination ingredients.
Because it takes these products to grow a virus or bacterium, other toxins can get in. These are called 'adventitious agents'.
Contaminants that have been known to get in include hepatitis B, simian virus 40 (a known cause of cancer), nvCJD and infectious retroviruses.
The FDA wrote:
'A discussion of adventitious agents and vaccines is pertinent in the context of emerging infectious diseases. Many novel vaccines are produced in animal cell substrates, and emerging infectious diseases may theoretically be transmitted from animals to humans through these vaccines. The challenge of identifying potential adventitious agents in vaccines closely parallels the challenge of identifying the agents causing particular emerging infectious diseases.
In the past, biologic products have served as vectors for viral diseases. Examples include the contamination of yellow fever vaccine with hepatitis B virus in the 1940s (because a human-derived excipient contained hepatitis B virus), contamination of early polio and adenovirus vaccines with simian virus 40 in the late 1950s and early 1960s, contamination of blood products with hepatitis viruses and HIV, and contamination of dura mater grafts with the Creutzfeldt-Jakob disease agent. In these examples, either human or animal materials used in production usually caused the contamination.
Production of viral vaccines generally involves inoculation of a cell substrate with a vaccine seed and purification of bulk product from these cells after a sufficient time for replication of the virus or production of vaccine proteins.
Other raw materials (e.g., tissue culture reagents, stabilizers) may be added to the product at various stages of production.
Thus, adventitious agents could theoretically enter a viral vaccine through any of these ingredients.'
(Adventitious Agents in Vaccines, FDA, 2001).
To try to counter this problem, vaccine plants use thimerosal, 2-phenoloxyethanol, polysorbate 80 or another toxic substance that is capable of killing all life including unwanted pathogens. This is why even so called 'mercury-free' vaccines cannot claim to be 100% mercury free. Trace amounts used in manufacture can end up in vaccines labelled mercury free and in any case, all vaccine plants use it so there is always the chance of contamination unless you have extremely expensive, high-tech facilities which would reduce profits.
The issue of advantitious agents and the need for powerful and poisonous disinfectants has been implicated in the development of autism and other disorders without even considering the actual added ingredients in vaccines.
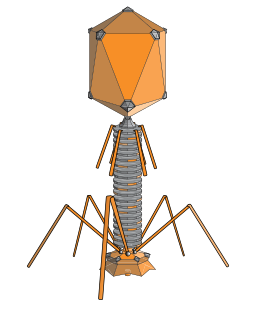
Contamination of Live Viral Vaccines with Bacteriophages
Live viral vaccines such as MMR, flu and chickenpox may also become contaminated with bacteriophages - viruses that replicate within a bacterium and can incorporate into your DNA. They use a syringe like motion to inject genetic material and once complete this is irreversible. Infection with these bacteriophages via vaccination not only occurs but could potentially cause permanent DNA damage.
Biologicals wrote:
'In 1973, live bacterial viruses (bacteriophages) were identified in several lots of live viral vaccines that had been submitted by Merck to the Bureau of Biologics (BB), Food and Drug Administration (FDA) for lot release.
Bacteriophages were reported as a contaminant of bovine sera in 1972 [23]. Follow-up studies by BB confirmed and extended those initial results [24]. Since some lots of sera contained over 104 pfu/ml of coliphage, it was reasonable to assume that bovine sera used in the manufacture of live viral vaccines might contain bacteriophages that could be carried through the manufacturing process into the final products.
Initial studies showed that the live viral vaccines from Merck contained bacteriophages at low concentrations (1–5 pfu/ml). The discovery was confirmed by an independent laboratory at BB using coded samples that included positive and negative controls along with samples of vaccine lots that had been reported positive in the initial experiments.
Soon after the results were confirmed by BB, Merck was contacted and asked to independently confirm the results using retention samples of the same lots of vaccines that were positive at BB. The manufacturer was initially unable to confirm the results. Discussions of the assay procedure did not reveal any differences that could explain the disparity in results. Personnel from BB went to the manufacturer's laboratory with all of their reagents and equipment in an attempt to replicate the results at the manufacturer's facility. Using the FDA procedure, the manufacturer's samples were shown to be positive for bacteriophages at the manufacturer's laboratory. The initial negative results had been due to the fact that the manufacturer's centrifugation step was different from that used at FDA, and it had not been sufficient to pellet the bacteriophages.
The initial findings were rapidly extended to other live viral vaccines and other manufacturers. The results showed that it was a general issue since 11 of 60 lots of live measles, mumps, rubella, and polio vaccines were positive for bacteriophages when tested using the C-3000 and K-12 strains of Escherichia coli as the host [25]. The host spectrum was expanded to include other bacterial strains, but no additional phages were detected. Only one phage, φV−1, was isolated from vaccines [26].'
However, regulatory authorities were more concerned that if they immediately banned the contaminated vaccines they would affect vaccine supply so they decided to continue to inject knowingly contaminated vaccine into the public, saying the risks of being injected with bacteriophages were only 'theoretical' whereas vaccines were 'safe'.As there is next to no research on the long-term impact of bacteriophages on health, immunity, cognitive function or any other physiological aspect, they really don't have much basis for saying vaccines are safe.
Biologicals wrote:
'In addition to the regulatory issues raised by bacteriophages in live viral vaccines, there was a question of whether the presence of viable phages in vaccines constituted a threat to the public health. The theoretical risks included both indirect and direct effects of phages on humans: (a) the induction of a toxin by phages in appropriate bacterial hosts followed by a disease that is due to the toxin such as diphtheria caused by corynebacteria and its phages; and (b) the induction of changes in human cells which could then lead to any of a variety of diseases.'
If autism does have a genetic component, how do authorities know that mass vaccination with bacteriophage contaminated vaccines does not alter the genome of vulnerable babies at critical stages of their development?
Retroviral Contamination
As many vaccines including MMR are cultured on chick embryo, they may become contaminated with infectious avian retroviruses. The Journal of Clinical Microbiology wrote:
'Vaccines are very effective at reducing death and suffering caused by viral diseases. Many vaccines are generated on primary avian cells, such as the vaccines for yellow fever, measles, and mumps. International health authorities require vaccines produced on avian cells to be tested for and demonstrated to be free of infectious avian retroviruses such as avian leukosis virus (ALV) and reticuloendotheliosis virus (REV) (1, 2, 3, 4). For example, detailed regulatory requirements are outlined specifically for the detection of ALV and REV in the European Pharmacopeia (Ph. Eur.) (2). Furthermore, the detection of reverse transcriptase (RT) activity, which is indicative of the presence of retroviruses, by highly sensitive PCR-based methods is recommended by several international regulatory authorities (3, 4, 5).
The detection of RT activity in yellow fever, measles, and mumps vaccines (6, 7) clearly demonstrated a need for effective methods to test for the presence of replication-competent retroviral contaminants. It was discovered upon further investigation that nonreplicative endogenous avian retroviral elements not associated with infectious retroviruses were responsible for the positive RT signals observed in these vaccines (8, 9, 10, 11, 12). As a consequence, it is now understood that it is necessary to test for the presence of infectious retroviral properties in addition to the presence of RT activity.'
Other retroviruses, such as those from primates and from mice have been found to contaminate vaccines that have been cultured on tissue from those species.
The xenotropic murine leukemia virus-related virus (XMRV) - a mouse leukaemia - has been found in human prostate cancers and in people suffering from chronic fatigue syndrome.
Frontiers in Microbiology wrote:
'One of the most widely distributed biological products that frequently involved mice or mouse tissue, at least up to recent years, are vaccines, especially vaccines against viruses. Some, for instance vaccines against rabies virus (Plotkin and Wiktor, 1978), yellow fever (YF) virus (Frierson, 2010), and Japanese encephalitis (JE) virus (Inactivated Japanese Encephalitis Virus Vaccine, 1993), consisted of viruses that were cultured on mouse brains. Such vaccines were in use from 1931 (YF vaccine) until now (JE vaccine, licensed in Japan since 1954). For rabies virus, early vaccines were mainly of goat or sheep nerve tissue origin. In addition, suckling mouse brain-derived rabies virus vaccines were used in South America and France (Plotkin and Wiktor, 1978). No mouse-derived rabies vaccine was ever licensed in the USA (Dennehy, 2001). Live-attenuated YF vaccines were originally also grown on mouse brain, but an YF vaccine grown on chicken eggs (named 17D) became available in 1937, and was since the vaccine of choice in the America's. In 1962, contamination of the 17D vaccine with oncogenic avian leukosis virus was detected both in England and in the USA, but fortunately no excess of cancer incidence among vaccines was reported (Frierson, 2010).'
Although being the most effective means to prevent infectious diseases and to safe lives, serious contamination problems involving vaccines have occurred (Pastoret, 2010). Contamination with unrelated viruses such as the presence of hepatitis B virus (HBV) in YF vaccine preparations stemming from the use of human serum for stabilization, and simian virus 40 (SV40) and foamy viruses through the use of monkey cell cultures (Pastoret, 2010). Some vaccine viruses are inactivated before use, hopefully also inactivating any contaminating virus particles, but the contaminating virus may be more stable than the vaccine virus. For instance, SV40 is highly resistant to inactivation (Murray, 1964). Endogenous retroviruses constitute a distinct class of contaminating viruses, as these viruses are encoded by all cells of a certain species, and therefore cannot be avoided even through rigorous screening (Miyazawa, 2010). Contamination with endogenous avian leukosis viruses is a major problem for vaccine viruses grown in chicken embryos or chicken embryonic fibroblasts (Hussain et al., 2003). Infectious cat endogenous RD-114 virus has been found in several veterinary vaccines produced in cat cell cultures (Miyazawa et al., 2010; Yoshikawa et al., 2010).'
Although no murine leukaemia has been isolated in autistic people, only one study of autistic people has been done to see if this is the case - on 25 subjects - and since the virus has only recently been discovered to infect humans, there is no way that scientists can know what the risks of XMRV are.
Other viruses and bacteria - such as polyoma viruses (viruses harmless in their host but harmful when injected into another species) have been found in the post-mortem brains of people with autism. Since everyone has a blood/brain barrier these viruses should not have been present in the brain. Polysorbate 80 - used in vaccines - is a transporter of drugs to the brain and is used specifically in medications such as antidepressants because it allows the drug direct access to the brain. Using such an ingredient in vaccines could allow vaccine viruses and contaminants to permeate the blood/brain barrier.
The Journal of Neurovirology wrote:
'Autism is a highly heritable behavioral disorder. Yet, two decades of genetic investigation have unveiled extremely few cases that can be solely explained on the basis of de novo mutations or cytogenetic abnormalities. Vertical viral transmission represents a nongenetic mechanism of disease compatible with high parent-to-offspring transmission and with low rates of disease-specific genetic abnormalities. Vertically transmitted viruses should be found more frequently in the affected tissues of autistic individuals compared to controls. Our initial step was thus to assess by nested polymerase chain reaction (PCR) and DNA sequence analysis the presence of cytomegalovirus (CMV), Epstein-Barr virus (EBV), herpes simplex virus type 1 (HSV1), herpes simplex virus type 2 (HSV2), human herpes virus 6 (HHV6), BK virus (BKV), JC virus (JCV), and simian virus 40 (SV40) in genomic DNA extracted from postmortem temporocortical tissue (Brodmann areas 41/42) belonging to 15 autistic patients and 13 controls. BKV, JCV, and SV40 combined are significantly more frequent among autistic patients compared to controls (67% versus 23%, respectively; P < .05). The majority of positives yielded archetypal sequences, whereas six patients and two controls unveiled single-base pair changes in two or more sequenced clones. No association is present with the remaining viruses, which are found in relatively few individuals (N <or= 3). Also polyviral infections tend to occur more frequently in the brains of autistic patients compared to controls (40% versus 7.7%, respectively; P = .08). Follow-up studies exploring vertical viral transmission as a possible pathogenetic mechanism in autistic disorder should focus on, but not be limited to, the role of polyomaviruses.'
The most recent contamination fiasco occured with the rotavirus gastroenteritis vaccine, when DNA from a pig virus, porcine circovirus type 1 and 2, was discovered in the vaccine.
Biologicals wrote:
'DNA from porcine circovirus type 1 (PCV1) and 2 (PCV2) has recently been detected in two vaccines against rotaviral gastroenteritis from manufacturers A and B. We investigated if PCV1 sequences are present in other viral vaccines. We screened seeds, bulks and final vaccine preparations from ten manufacturers using qRT-PCR. We detected 3.8 × 10³ to 1.9 × 10⁷ PCV1 DNA copies/milliliter in live poliovirus seeds for inactivated polio vaccine (IPV) from manufacturer A, however, following inactivation and purification, the finished IPV was PCV1-negative. PCV1 DNA was not detectable in live polio preparations from other vaccine producers. There was no detectable PCV1 DNA in the measles, mumps, rubella and influenza vaccines analysed including material supplied by manufacturer A. We confirmed that the PCV1 genome in the rotavirus vaccine from manufacturer A is near full-length. It contains two mutations in the PCV cap gene, which may result from viral adaptation to Vero cells. Bulks of this vaccine contained 9.8 × 10¹⁰ to 1.8 × 10¹¹ PCV1 DNA copies/millilitre and between 4.1 × 10⁷ and 5.5 × 10⁸ DNA copies were in the final doses. We found traces of PCV1 and PCV2 DNA in the rotavirus vaccine from manufacturer B. This highlights the issue of vaccine contamination and may impact on vaccine quality control.'
The virus wasn't meant to be there and had occured in the initial manufacture of the product. Despite this, the FDA put the infected vaccine back on the market and into innocent children without trying to re-formulate the vaccine or remove the virus from it - again, putting vaccine supply and profit before safety.
In the description of the vaccine, the manufacturer wrote:
'(PCV-1) is present in ROTARIX. PCV-1 is not known to cause disease in humans.'
Circoviruses cause muscle wasting, respiratory disease, immune system damage, infertility and sometimes death. Although the disease isn't known to infect humans, they can't guarantee it doesn't and they have no idea of the effect of introducing pig virus DNA into the human genome.
How can health authorities say that the benefits outweigh the risks when they didn't even know the porcine circovirus was in there?
1. Adventitious Agents and Vaccines, Philip R Krause, FDA, 2001
Adventitious agents in viral vaccines: Lessons learned from 4 case studies
http://commons.wikimedia.org/wiki/File:PhageExterior.svg
Association of autism with polyomavirus infection in postmortem brains
Of Mice and Men: On the Origin of XMRV
Porcine circoviruses and rotavirus vaccines
Investigation of porcine circovirus contamination in human vaccines
Other pages:
This is the text-only version of this page. Click here to see this page with graphics.
Edit this page |
Manage website
Make Your Own Website: 2-Minute-Website.com